GENERAL INFORMATION (WHAT IS ANTIFREEZE?)
|
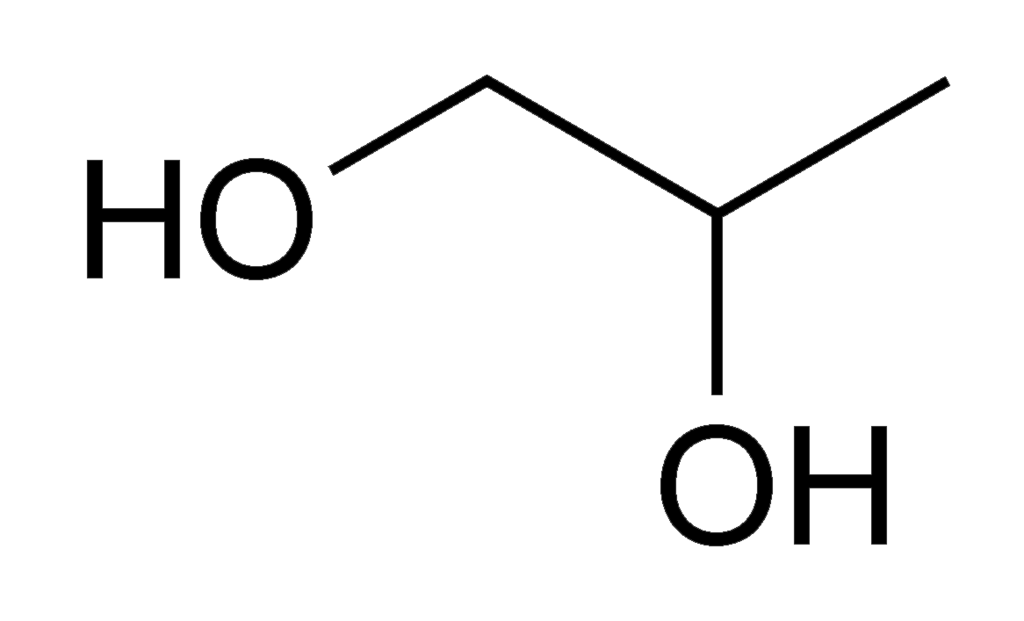
Above: The cell structure for Ethylene Glycol
Below: The cell structure for Propylene Glycol |
The paragraph above means the main purpose of antifreeze is to keep a car's fluids from overheating while driving or freezing while not in use during the winter or in a cold environment. It can also be used as a windshield wiper fluid. Antifreeze, with a chemical formula of C2H4O + H2O → HO–CH2CH2–OH, has 6 carbon cells, 12 hydrogen cells, and 4 oxygen cells. Antifreeze can also be found as propylene glycol (the non-poisonous version). The boiling point of antifreeze is 197°C (387°F), and it has a freezing point of -55°C (-67° F). The colour of antifreeze can be anything, but it is most commonly orange or green. Antifreeze normally comes in a liquid, obviously. Antifreeze is flammable at a temperature of 115°C/210°F. |